
Stents at UTMC improve the lives of patients
Approved device reopens blocked arteries in legs
3/25/2013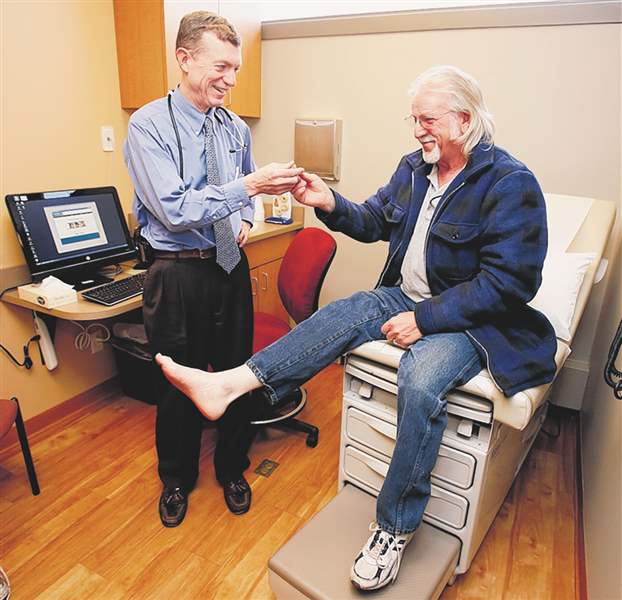
Dr. Mark Burket, UTMC chief cardiologist, examines Keith Bowman of Cincinnati, who has peripheral artery disease.
The Blade/Jeremy Wadsworth
Buy This Image

Dr. Mark Burket, UTMC chief cardiologist, holds the Zilver stent for patients with peripheral artery disease.
A small, mesh cylindrical device that looks like the metal spirals that hold together notebook paper has garnered the University of Toledo Medical Center recognition and is improving the lives of patients.
The Zilver PTX Vascular Stent was approved by the U.S. Food and Drug Administration in November to reopen blocked arteries in the legs of patients who suffer from peripheral artery disease, or PAD. In January, the stent was placed in the right thigh artery of Cincinnati-area resident Keith Bowman, 64. The first to receive the device, Mr. Bowman only has good things to say about the way his leg feels.
“I used to only be able to walk a half block, and now I can walk two miles,” he said, adding that he was down for only a couple of days after the procedure. “After the pain from the operation, it’s been great.”
The stent bends as Mr. Bowman moves his leg, said Dr. Mark Burket, chief of cardiovascular medicine at UTMC. As the principal investigator in the study of the stent at the former Medical College of Ohio, which enrolled 25 patients in the study, Dr. Burket said Mr. Bowman had pain in his leg when he walked. The pain was a result of claudication, which is the blockage of blood flow to an arm or leg.
“Also referred to as PAD, essentially the artery that goes down the thigh was plugged and had renarrowed,” as a result of a buildup of cholesterol, Dr. Burket said.
The simple and safe procedure involves using a balloon to expand the artery and then the stent is slipped into the artery. Though it’s an outpatient procedure, Mr. Bowman stayed at the hospital overnight because of his long drive home.
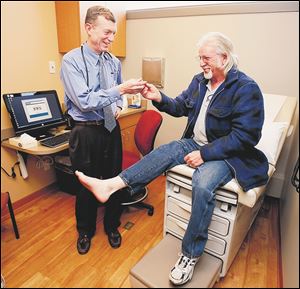
Dr. Mark Burket, UTMC chief cardiologist, examines Keith Bowman of Cincinnati, who has peripheral artery disease.
“What’s neat about the new [stent] that we just got – he was the first patient to get it – is that it comes with a special drug coating that decreases the chance of narrowing by about 50 percent,” said Dr. Burket said.
During the five years of research, UTMC was the largest U.S. enrolling site for the study among university hospitals, Dr. Burket said. The hospital was one of the first in the nation to obtain access to the device, described as giving new life to patients who once endured constant pain and allowed others who could not walk to do so again.
In 2009, Mr. Bowman had a bypass operation on his left leg, which functions, but not as well as the right leg does now since getting the new stent. Doctors previously tried to open the artery on the right leg, but it wasn’t successful because it narrowed again. Mr. Bowman learned about the vascular stent research study here from his Hamilton, Ohio, cardiologist who was aware of it because he once practiced in this region. About 10 other patients have received vascular stents since Mr. Bowman, and each has been successful, Dr. Burket said.
“I would rather have this than another bypass. My right leg is so much better than the left leg,” Mr. Bowman said. “This surgery I had on the left leg is no fun. I can tell the difference in the flow.”
Contact Rose Russell at: rrussell@theblade.com or 419-724-6178.