
Tobacco comapnies win challenge against graphic cigarette package labels
Appeals court blocks new law
8/25/2012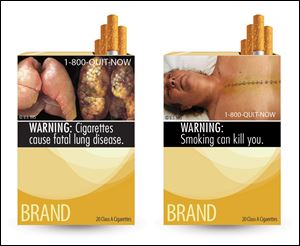
Two of nine new warning labels cigarette makers would have had to use by the fall of 2012.
Tobacco companies defeated a U.S. law forcing cigarette packaging and advertisements to display images such as diseased lungs, persuading a federal appeals court that the requirements violate their free speech rights.
In a 2-1 decision, the U.S. Court of Appeals in Washington today ruled that Food and Drug Administration regulations mandating visual-image warnings of smoking’s health risks, along with the telephone number 1-800-QUIT-NOW, are “unabashed attempts to evoke emotion” and “browbeat consumers” to stop buying the companies’ products.
“These inflammatory images and the provocatively named hotline cannot rationally be viewed as pure attempts to convey information to consumers,” U.S. Circuit Judge Janice Rogers Brown wrote in her majority opinion.
Commonwealth Brands Inc., Liggett Group LLC and Santa Fe Natural Tobacco Co. sued the FDA last year, claiming the mandates for cigarette packages, cartons and advertising, passed as part of the Family Smoking Prevention and Control Act, violated the First Amendment.
The companies said in court papers that complying with the requirements would cost them a total of about $20 million. The mandate, scheduled to go into effect next month, was put on hold by a lower-court judge while the appeals court considered its legality.
The government argued in court papers that nine images selected by the agency to be placed on packages and advertisements are true depictions required by Congress in the law to show the negative health consequences of smoking.
The graphics were supposed to cover the top half of the front and back of cigarette packages and 20 percent of print advertisements. The FDA estimated the visual warnings would help lower the smoking rate by about 0.212 percentage points, according to the lower court judge who also ruled against the FDA.
Jennifer Haliski, an FDA spokeswoman, said the agency doesn’t comment “on possible, pending or ongoing litigation.”
The Campaign for Tobacco-Free Kids, an anti-smoking group, urged the government to appeal the ruling, noting that a federal appeals court Cincinnati upheld the packaging requirements in March.
“The split decisions make it likely the U.S. Supreme Court will settle the issue,” the group said in an e-mailed statement.
During a Feb. 1 hearing before U.S. District Judge Richard Leon, Mark Stern, a Justice Department lawyer, compared the FDA’s proposed cigarette warnings to those on charcoal that advise people to not use it indoors, noting that 28 people a year die from carbon monoxide poisoning from using charcoal inside their homes.
With cigarettes, there are 440,000 deaths, Stern said.
“It’s very unusual to sell a product that when used as intended will kill you,” Stern said.
He said that the image of a man with cigarette smoke coming out of a tracheotomy hole in his throat conveys addictiveness.
Brown said the image could be misinterpreted to suggest that the procedure is a common consequence of smoking.
In a dissent, U.S. Circuit Judge Judith Rogers said Brown’s opinion applied the wrong level of First Amendment scrutiny and disregarded the tobacco companies’ history of deceptive advertising.
Noel Francisco, a lawyer for R.J. Reynolds, a unit of Reynolds American Inc., argued that “the purpose of the warnings is not to inform, but to scare consumers into adopting the government’s course of action.” He said during the February hearing that the government was using “threats and fear” to motivate people to stop using a lawful product.
FDA “failed to present any data” showing that the proposed graphic warnings “will accomplish the agency’s stated objective of reducing smoking rates,” Brown said in the opinion.
“The First Amendment requires the government not only to state a substantial interest justifying a regulation on commercial speech, but also to show that its regulation directly advances that goal,” she said in the ruling.
On Aug. 15, the High Court of Australia upheld that country’s requirement that cigarettes be sold in uniform packages barring display of company trademarks. New Zealand and the U.K. are among countries whose governments have indicated interest in implementing similar legislation, which takes effect in Australia Dec. 1.
The case is R.J. Reynolds Tobacco Co. v. U.S. Food and Drug Administration, 11-5332, U.S. Court of Appeals for the District of Columbia (Washington).